Contact UsCONTACT
Please feel free to contact us if you have any questions or concerns.
Inquiry FormStories
STORIES
SERIES EMBARK

FIMECS, Inc. is a bio-venture that aims to create innovative drugs for targets previously considered undruggable by using targeted proteolysis inducers, a novel modality for drug discovery. It was established in January 2018 by researchers from Takeda Pharmaceutical Company Limited ("Takeda") using the company's venture establishment support program. We interviewed Yusuke Tominari, co-founder and CEO, and Kanae Gamo, co-founder and CSO, about the background of the start-up and the uniqueness of the established target protein degradation inducer platform. (Interviewer: Katsuyoshi Masuda)
I joined Takeda in 2006 after working in the Department of Synthetic Natural Products Chemistry at the Graduate School of Pharmaceutical Sciences of the University of Tokyo. I worked in the Cancer Research Division in the research and development of anti-cancer drugs, and later in the Inflammation Research Group in drug discovery for immunological diseases. In this context, since around 2014, I have been active in a volunteer working group "F-iMec (Future immunology-Unit Medicinal Chemistry)" to validate research ideas with a long-term perspective and impact.
In the WG, I was initially working on the theme of how to make drugs from so-called "Undruggable Targets," which are considered to be difficult to discover using existing drug discovery technologies. In that research, I focused on a drug target called IRAK-M, which could be used for cancer immunology. However, IRAK-M belongs to a protein called pseudokinase, and it turned out to be difficult to inhibit its function with the binder I had obtained, so I stopped the research early. On the other hand, since the technology of target protein degradation inducers was beginning to attract attention, we were considering it as the next theme to work on. We thought that we could make it into a drug by combining it with the binder of IRAK-M that we had already acquired, and proposed it as a project after initial discussions.
Once evaluated as a top priority, but just then the R&D structure was changed and R&D in oncology, including cancer immunology, was centralized in Boston, USA. At that time, the Shonan Research Center was to focus mainly on central nervous system diseases, and medicinal chemists were at a crossroads: stay at the company and work on central nervous system diseases, change jobs to other pharmaceutical companies, or take advantage of the venture establishment system offered by Takeda and start a company to continue research and development. He found a colleague to co-found the company, so he chose to start his own business through Takeda's Entrepreneurial Venture Program, a program that supports the establishment of a venture company.

Because my father was a biologist, I majored in biology with a particular interest in life phenomena and the origins of disease. After graduating from the Graduate School of Pharmaceutical Sciences at Osaka University, I joined Takeda in 2009 and have been engaged in biological research with a single-minded desire to create cancer drugs.
I was also involved in WG as a Biologist, but as Tominari mentioned, there was a major reorganization of the research department, and according to the policy at that time, the Shonan Research Institute could not conduct drug discovery research in the field of cancer.... If I had been single, I would have gone to Boston, but I could not take the plunge considering my family and career. On the other hand, I was also concerned that even if I moved to another pharmaceutical company, the same thing would likely await me due to organizational reforms and policy changes. Since Takeda had given us an option that would allow us to formulate our own strategy and reap the fruits of the research we wanted to pursue, I felt that I would regret not taking advantage of it, and so I chose the path of co-founder.
In order to receive investment from Takeda through the venture establishment support program, we also had to focus on obtaining investment from outside of Takeda. We visited many venture capital firms, but at the time, many were skeptical of our technology and were reluctant to invest. We were able to start the company after negotiations were concluded just before the deadline, and the investment decision was made in just three days. The founding members of the company started with two WG members in addition to Gamo, but it was a tough time for us because we had to continue our research while also continuing our fundraising work because we were in a phase where competitors were launching their products overseas.
After our presentation at the conference at the end of 2018, we were contacted by a professor from the Kyoto University Graduate School of Medicine, who discussed the possibility of creating an epoch-making new drug by crossing our technology with a compound he was researching, and a joint research project on a new drug for cancer was finalized. Around that time, we also began discussions with Kyoto-iCAP and were able to obtain their investment. The proteolysis inducer we were developing was recognized for its potential as a new treatment for patients who lack treatment options, and we were able to raise funds from Kyoto-iCAP, the University of Tokyo's IPC (University of Tokyo Collaborative Platform Development Corporation), and funds from ANRI.

Our goal is to create innovative drugs for targets that have been considered difficult to discover by using a new drug discovery approach called Targeted Protein Degradation (TPD). When a protein called E3 ligase, which marks an abnormal protein to be degraded (target), binds to the target through protein-protein interaction (PPI), ubiquitin is transferred to the target, and the ubiquitin is used as a marker for the target. The enzyme proteasome then degrades the target protein. The approach is to realize this with a compound. However, it is clear that if the E3 ligase changes, the protein to be degraded also changes, or the target of degradation changes depending on the way the interaction takes place. This is because protein-protein interactions are greatly affected by the compatibility of the target protein and the E3 ligase. In order to degrade a target protein, it is necessary to find the optimal combination of E3 ligase, compound binding site, and strength of protein-protein interaction from a myriad of combinations. We concluded that it would be impossible at this stage to design the optimal combination using computational chemistry, so we decided to create a unique drug discovery platform.
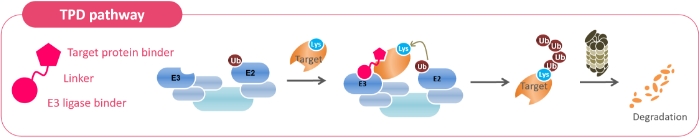
The proprietary Rapid Protein Proteolysis Inducer Discovery System (RaPPIDS) is based on Diversity-Oriented Synthesis (DOS: DOS), which efficiently creates a variety of compounds with different molecular skeletons. It is based on Diversity-Oriented Synthesis (DOS), which efficiently creates a variety of compounds with different molecular backbones, and consists of two stages: creation of compounds that show activity against drug targets (lead compounds) and optimization of the obtained lead compounds. The main feature of this system is that it enables automated synthesis of compounds, which enables efficient creation of optimal pharmaceutical compounds in a short period of time, which is a differentiating advantage of this system.
We have been in full-scale development since our founding and have now reached the stage where we can synthesize more than 1,000 target proteolysis inducers in a week utilizing automated synthesis. This has allowed us to explore over 600 E3 ligases in total, while there are only around 10 reported E3 ligases that have been used as target proteolysis inducers. ligase, but if it is found to be degradative, it can be used as a lead compound or a tool compound to determine if it is a suitable target for drug discovery. Therefore, compared to conventional methods, this method is characterized by its low cost and early Go/No Go decision. On the other hand, it is necessary to optimize the compound while identifying the E3 ligase that contributes to degradation.
In major pharmaceutical companies, for example, it takes about five researchers about one year to create 200-300 compounds in the optimization phase. It is common for optimization to proceed through repeated evaluations and validations of these compounds in parallel. A similar process is required in the lead discovery phase, and even if parallel synthesis is used, it is difficult to synthesize 1,000 compounds per week. We have realized low-cost and short lead time for synthesis of multiple samples by automated synthesis, which is time-consuming and costly. Even if the synthesized compound has no activity, the results are known in 2 to 3 months, which has the advantage of allowing us to stop the project early and focus on another project.
In-house pipeline research is being conducted on several pipeline proteins, including IRAK-M, a protein that causes cancer immunosuppression in non-small cell lung cancer and pancreatic cancer, and TRIB1, a protein that targets acute myeloid leukemia as a target disease.
Both of these two target proteins belong to the class of pseudokinases, which have been considered undruggable. Pseudokinases are also called pseudokinases and have been considered undruggable by conventional drug discovery technologies because they do not have kinase activity. However, by combining Pseudokinase with the technology of target protein degradation inducers, it is a compatible target class that can be made into drugs. We are not focusing only on pseudokinases, but they are a target class that has been evaluated as being compatible with target proteolysis inducers as one approach, and we are developing drugs for them in-house.
Meanwhile, RaPPIDS is being used to build a joint development pipeline through collaborations with pharmaceutical companies. One such collaboration, announced on February 1, 2022, is with Astellas Pharma Inc. for targeted proteolysis inducers against multiple targets. We were able to initiate the collaboration due to high expectations for our proprietary E3 ligase binders (including E3 ligases with tissue-selective and cancer-selective expression) and efficient target proteolysis inducer synthesis technology. We believe that the collaborative research program is an important business model to demonstrate that we can continue to generate valuable assets from our platform.
Ventures in the U.S. and other countries have great financial power, and not only leading ventures but even some late-stage ventures have already raised funds through IPOs. In order to compete with these companies, we need to obtain large deals through joint research projects using our platform. To do so, it is important how we increase the value of our platform, and we must further refine it technologically.
At the same time, we would like to contribute to the revitalization of research and development of targeted protein degradation inducers in Japan and Asia. As part of this effort, we plan to hold the 2nd Targeted Protein Degradation Conference in Japan on July 26-27, 2023, at the Shonan This will be the second conference following the one in 2019, and with half of the 22 speakers being executive class from overseas, we expect that cutting-edge research results will be shared with participants and that research on targeted protein degradation inducers in Japan will be further stimulated.
This is for people who are involved in drug discovery research, but if you have interesting assets of your own, I recommend that you should try to start your own business. This is subject to the approval of your current company, but if you don't start up when you have the chance, you will never have the opportunity again. If there is an opportunity for a spin-off, I think you should not think about failure after starting a business, but rather go forward with an image of what you can do to make it a success.
I am the type of person who makes decisions based on my senses, but I have a policy that the person I can ultimately trust is myself. It is natural that starting a business involves risk, but I believe in myself, and even if I fail, it will be my own responsibility. However, I cannot do it alone, so I think it is important to have friends who share my ideas.
(Interviewed in May 2023. Affiliations, positions, etc. are as of the time of the interview)
FIMECS is a drug discovery venture company that conducts research and development of novel drugs with proteolysis as their mechanism of action. FIMECS aims to target proteins that have been considered difficult to discover in drug development to date using its proprietary drug discovery platform. The enthusiasm and sense of mission of CEO Tominari and CSO Gamo for this technology has attracted a large number of employees interested in the technology. Kyoto-iCAP will continue to support FIMECS's activities and hopes that the innovative new drugs it develops will benefit healthcare and patients.

Hiroyuki Ueno

FIMECS, Inc. Website
Please feel free to contact us if you have any questions or concerns.
Inquiry Form